Abstract
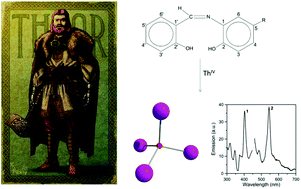
Thorium(IV) complexes are currently attracting intense attention from inorganic chemists due to the development of liquid-fluoride thorium reactors and the fact that thorium(IV) is often used as a model system for the study of the more radioactive Np(IV) and Pu(IV). Schiff-base complexes of tetravalent actinides are useful for the development of new separation strategies in nuclear fuel processing and nuclear waste management. Thorium(IV)–Schiff base complexes find applications in the colorimetric detection of this toxic metal ion and the construction of fluorescent on/off sensors for Th(IV) exploiting the ligand-based light emission of its complexes. Clusters of Th(IV) with hydroxide, oxide or peroxide bridges are also relevant to the environmental and geological chemistry of this metal ion. The reactions between Th(NO3)4·5H2O and N-salicylidene-o-aminophenol (LH2) and N-salicylidene-o-amino-4-methylphenol (L′H2) in MeCN have provided access to complexes [Th4O(NO3)2(LH)2(L)5] (1) and [Th4O(NO3)2(L′H)2(L′)5] (2) in moderate yields. The structures of 1·4MeCN and 2·2.4 MeCN have been determined by single-crystal X-ray crystallography. The complexes have similar molecular structures possessing the {Th4(μ4-O)(μ-OR′)8} core that contains the extremely rare {Th4(μ4-O)} unit. The four ThIV atoms are arranged at the vertexes of a distorted tetrahedron with a central μ4-O2− ion bonded to each metal ion. The H atom of one of the acidic –OH groups of each 3.21 LH− or L′H− ligand is located on the imine nitrogen atom, thus blocking its coordination. The ThIV centres are also held together by one 3.221 L2− or (L′)2− group and four 2.211 L2− or (L′)2− ligands. The metal ions adopt three different coordination numbers (8, 9, and 10) with a total of four coordination geometries (triangular dodecahedral, muffin, biaugmented trigonal prismatic, and sphenocorona). A variety of H-bonding interactions create 1D chains and 2D layers in the crystal structures of 1·4 MeCN and 2·2.4 MeCN, respectively. The structures of the complexes are compared with those of the uranyl complexes with the same or similar ligands. Solid-state and IR data are discussed in terms of the coordination mode of the organic ligands and the nitrato groups. 1H NMR data suggest that solid-state structures are not retained in DMSO. The solid complexes emit green light at room temperature upon excitation at 400 nm, the emission being ligand-centered.
- This article is part of the themed collections: Spotlight Collection focused on Inorganic Chemistry in Greece and Modern coordination chemistry


 Please wait while we load your content...
Please wait while we load your content...