Mononuclear manganese(iii) complexes with reduced imino nitroxide radicals by single-electron transfer and intermolecular hydrogen bonds as an intramolecular structural driving force†
Abstract
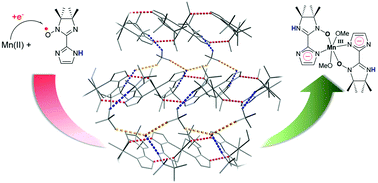
Manganese(III) complexes were synthesized by one-electron transfer from a Mn(II) ion to the imino nitroxide radical 2-(2-imidazolyl)-4,4,5,5-tetramethylimidazoline-1-oxyl (IMImH) in methanol. After the manganese ions attained the +III oxidation state, the imino nitroxide radicals were found to be irreversibly reduced in the complexes. Depending on the synthesis conditions, two complexes differing by their counter-anions were isolated as single crystals. These are [Mn(IMHIm)2(MeOH)2]ClO4·H2O (1) and [Mn(IMHIm)2(MeOH)2]PF6 (2), which crystallize in the monoclinic P21/n and triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space groups, respectively. The two complexes show Jahn–Teller distortions typical of Mn(III) centres and only reduced radicals are coordinated, as indicated by the N–O bond lengths and electroneutrality. In addition, the crystal structure analyses reveal two intermolecular hydrogen bonding networks. One involves counter-anions, water molecules and reduced radicals, and the other involves coordinated methanol molecules and imidazole moieties. These intermolecular interactions are driving forces that stabilize the two complexes. They also suggest that the tautomer is in the amino imine-oxide form after reduction of the radical and reveal the deprotonation of the imidazole ring, which is required for electroneutrality. This assessment is supported by single-crystal X-ray diffraction, EPR and Raman spectroscopy as well as magnetic and electrochemical studies.
space groups, respectively. The two complexes show Jahn–Teller distortions typical of Mn(III) centres and only reduced radicals are coordinated, as indicated by the N–O bond lengths and electroneutrality. In addition, the crystal structure analyses reveal two intermolecular hydrogen bonding networks. One involves counter-anions, water molecules and reduced radicals, and the other involves coordinated methanol molecules and imidazole moieties. These intermolecular interactions are driving forces that stabilize the two complexes. They also suggest that the tautomer is in the amino imine-oxide form after reduction of the radical and reveal the deprotonation of the imidazole ring, which is required for electroneutrality. This assessment is supported by single-crystal X-ray diffraction, EPR and Raman spectroscopy as well as magnetic and electrochemical studies.


 Please wait while we load your content...
Please wait while we load your content...