Luminescent anticancer ruthenium(ii)-p-cymene complexes of extended imidazophenanthroline ligands: synthesis, structure, reactivity, biomolecular interactions and live cell imaging†
Abstract
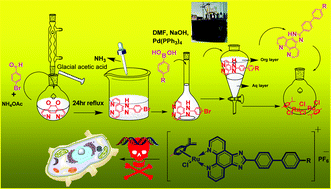
Of late, cancer has become a terrible disease affecting people throughout the world. Keeping this in mind, we tried to design drugs that are more lipophilic, target-specific, water-soluble, cytoselective and fluorescent. In this regard, we reported novel ruthenium(II)-p-cymene imidazophenanthroline scaffolds as effective DNA targeting agents. The planarity of imidazophenanthroline ligands caused the Ru(II) complex to be a good intercalator. An extended π-electronic conjugation was introduced in the imidazophenanthroline moieties through the Suzuki and Sonogashira coupling reactions. Here, we synthesized nine Ru(II) complexes (16a–b, 17a–d, and 19a–c). Among these, [(η6-p-cymene)RuCl(K2-N,N-2-(4′-methyl-[1,1′-BIphenyl]-4-yl)-1H-imidazo[4,5-f][1,10]phenanthroline)]·PF6 (16b) exhibited the best potency and selectivity with excellent cellular uptake; [(η6-p-cymene)RuCl(K2-N,N-2-(4-(phenylethynyl)phenyl)-1H-imidazo[4,5-f][1,10]phenanthroline)]·PF6 (17a) acted as a cytoselective probe for live cell imaging.


 Please wait while we load your content...
Please wait while we load your content...