Effect of the change of the ancillary carboxylate bridging ligand on the SMM and luminescence properties of a series of carboxylate-diphenoxido triply bridged dinuclear ZnLn and tetranuclear Zn2Ln2 complexes (Ln = Dy, Er)†
Abstract
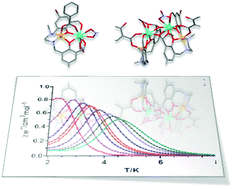
Eleven new dinuclear and tetranuclear compounds of general formulae [Zn(μ-L)(μ-X)Ln(NO3)2]·nS, [Zn2Dy2(μ3-L′)2(μ-sal)2(NO3)(CH3OH)](NO3)·5CH3OH and [Zn2Er2(μ3-L′)2(μ-sal)2(CH3OH)2](NO3)2·4CH3OH (X = benzoate, anthracenate, diclofenac, salicylate, 2,6-dihydroxybenzoate; Ln = Dy, Er; S = water, acetonitrile, methanol) were prepared from the N,N′-dimethyl-N,N′-bis(2-hydroxy-3-formyl-5-bromobenzyl)ethylenediamine compartmental ligand (H2L). Complexes 1–6 and 9–11 consist of diphenoxido-carboxylate triply bridged compounds, which differ mainly in the carboxylate bridging ligand. It should be noted that the acidic character of the salicylic acid promotes, in the presence of methanol, the methoxylation of the H2L ligand thereby yielding a hemiacetal H3L′, which is able to connect the Ln(III) ions of two ZnLn dinuclear units forming the Zn2Ln2 tetranuclear complexes 7 and 8. All compounds display SMM behaviour in the presence of an external field with effective energy barriers (Ueff) as high as 61 K. Magneto-structural data for these complexes reveal that their SMM behaviour is not only significantly affected by the type of Ln(III) ion but also by the carboxylate bridging ligand connecting the Zn(II) and Ln(III) ions. Photoluminescence properties have also been accomplished, showing that the ligands are able to sensitize lanthanide centred emissions in the visible and near-infrared regions with variable capacity. Moreover, the analysis of the luminescence decay curves reveals emission lifetimes in the range of few microsecond or hundreds of nanoseconds for Dy(III)-based or Er(III)-based luminophores, respectively.


 Please wait while we load your content...
Please wait while we load your content...