CoOx(OH)y/C nanocomposites in situ derived from Na4Co3(PO4)2P2O7 as sustainable electrocatalysts for water splitting†
Abstract
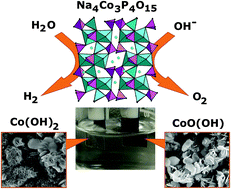
The great interest in developing efficient and stable bifunctional electrodes for electrocatalytic water splitting is due to the rapid growth in demand for sustainable and renewable energy sources. Here, we present an original electrode design strategy which can be used for the oxygen evolution reaction (OER) and the hydrogen evolution reaction (HER). This strategy is based on the direct formation of active catalytic material on the electrode surface during electrolysis. In our case, cobalt hydroxides (β-Co(OH)2 and CoO(OH) for HER and OER, respectively) in a carbon matrix are produced by the chemical and electrochemical transformation of various forms (glass, glass-ceramic and polycrystalline) of Na4Co3(PO4)2P2O7 during electrolysis in 1 M NaOH solution. The CoOx(OH)y/C composites demonstrate remarkable stability over time (more than 50 h), and bifunctional catalytic activity with an overall water splitting potential close to 1.8 V at a current density of 10 mA cm−2. The range of overpotentials for OER and HER is 346–365 and 326–369 mV, respectively. These electrodes can invert the OER and HER processes with a total overpotential of 0.6 V in the cell. This remarkable CoO(OH) ↔ Co(OH)2 inversion in the carbon matrix its further utility for creating bifunctional electrodes for long-life water splitting cells.


 Please wait while we load your content...
Please wait while we load your content...