A novel bis-1,2,4-benzothiadiazine pincer ligand: synthesis, characterization and first row transition metal complexes†
Abstract
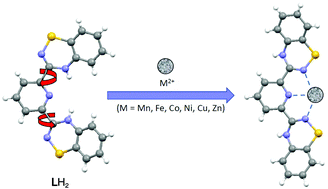
Reaction of 2,6-dicyanopyridine with 2 equiv. of 2-(propylthio)benzenamine in the presence of lithium bis(trimethylsilyl)amide, followed by ring-closing oxidation with N-chlorosuccinimide affords the novel tridentate ligand, 2,6-bis-(1′,2′,4′-benzothiadiazinyl)pyridine (LH2). Electrochemical studies on the free ligand LH2 reveal a single well-defined 2e− oxidation process with E1/2 = +0.90 V. EPR studies of the in situ chemical oxidation of LH2 reveal the generation of a benzotriazinyl radical. Reactions of ligand LH2 with a range of divalent transition metal salts in either MeOH or MeCN in a 2 : 1 ratio at ambient temperatures afforded mononuclear complexes with general formula [M(LH2)2][X]2 (M = Mn, X = CF3SO3 (1); Fe, X = CF3SO3 (2); Fe, X = BF4 (3); Co, X = Cl (4); Ni, X = Cl (5); Zn, X = CF3SO3 (6)) and the 1 : 1 complex [Cu(LH2)(NO3)2] (7). In all cases the LH2 ligand binds in a tridentate N,N′,N′′ chelate fashion via benzothiadiazinyl NBTDA and pyridyl Npy atoms. The low spin FeII complexes (2 and 3) were implemented for NMR and UV-Vis solution studies of ligand reactivity as well as cyclic voltammetry which reveal two 1e-oxidation waves. The metal complexes 1–6 are discussed and reveal a range of geometries between octahedral and trigonal prismatic with the greatest deviation from octahedral symmetry apparent for ions with no crystal field stabilisation energy, i.e. d10 ZnII and high spin d5 MnII ions.
- This article is part of the themed collection: Frontiers in coordination chemistry and its applications


 Please wait while we load your content...
Please wait while we load your content...