Elucidating white light emissions in Tm3+/Dy3+ codoped polyoxometalates: a color tuning and energy transfer mechanism study†
Abstract
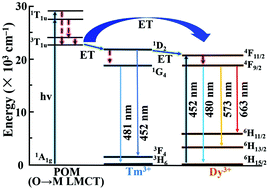
The double-tartaric bridging Tm-substituted POM derivative [N(CH3)4]6K3H7[Tm(C4H2O6)(α-PW11O39)]2·27H2O (1) was successfully synthesized and well characterized by various physico-chemical analyses. Furthermore, the mixed Dy3+/Tm3+ ion-based POM derivatives [N(CH3)4]6K3H7[DyxTm1−x(C4H2O6)(α-PW11O39)]2·27H2O (3–8) were first synthesized and confirmed by PXRD and IR spectra, indicating compounds 3–8 are isomorphic with 1. The detailed analyses of Ln–O–W bond angle and coordinated aqua ligands around emitting Ln3+ ions have revealed that the mentioned negative factors do not effectively affect the luminescence of emitting Ln3+ ions in 1–8. Investigations of PL emissions reveal that 3–8 can display color-tunable PL properties, emitting color from blue to white to yellow. The study of time-resolved emission spectroscopy of 6 indicates the energy can transfer from the LMCT excited state of POM fragments to Tm3+ and/or Dy3+ ions. Furthermore, the decreased luminescent lifetime of Tm3+ ions in 3–8 reasonably verifies the energy transfer from Tm3+ to Dy3+ ions to efficiently facilitate emissions of the Dy3+ centre.


 Please wait while we load your content...
Please wait while we load your content...