Abstract
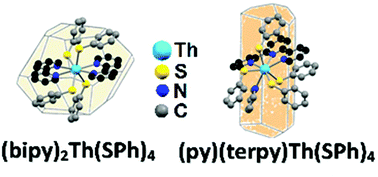
Thorium chalcogenolates Th(ER)4 react with 2,2′-bipyridine (bipy) to form complexes with the stoichiometry (bipy)2Th(ER)4 (E = S, Se; R = Ph, C6F5). All four compounds have been isolated and characterized by spectroscopic methods and low-temperature single crystal X-ray diffraction. Two of the products, (bipy)2Th(SC6F5)4 and (bipy)2Th(SeC6F5)4, crystallize with lattice solvent, (bipy)2Th(SPh)4 crystallizes with no lattice solvent, and the selenolate (bipy)2Th(SePh)4 crystallizes in two phases, with and without lattice solvent. In all four compounds the available volume for coordination bounded by the two bipy ligands is large enough to allow significant conformational flexibility of thiolate or selenolate ligands. 77Se NMR confirms that the structures of the selenolate products are the same in pyridine solution and in the solid state. Attempts to prepare analogous derivatives with 2,2′,6′,2′′-terpyridine (terpy) were successful only in the isolation of (terpy)(py)Th(SPh)4, the first terpy compound of thorium. These materials are thermochromic, with color attributed to ligand-to-ligand charge transfer excitations.
- This article is part of the themed collection: In celebration of Richard Andersen’s 75th birthday


 Please wait while we load your content...
Please wait while we load your content...
