A bromo-capped diruthenium(i,i) N-heterocyclic carbene compound for in situ bromine generation with NBS: catalytic olefin aziridination reactions†‡
Abstract
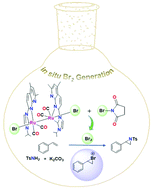
A bromo-capped metal–metal bonded diruthenium(I,I) complex Ru2(CO)4(PIN)2Br2 (1) (PIN = 1-isopropyl-3-(5,7-dimethyl-1,8-naphthyrid-2-yl)imidazol-2-ylidene) generates bromine with N-bromosuccinimide (NBS) at room temperature. Cycloalkene and stilbene are readily brominated by stoichiometric reactions with 1 and NBS. An analysis of the dibrominated products suggests the formation of cyclic bromonium intermediates indicating in situ Br2 generation. Complex 2, an iodide analogue of 1, is also synthesized. The reaction of 2 with N-iodosuccinimide releases I2, which is confirmed by the starch-iodine test. The catalytic utility of 1 is examined for the bromination of phenol. Catalyst 1, in combination with NBS and base, exhibits regioselectivity towards monobrominated products. Furthermore, efficient olefin aziridination is demonstrated utilizing catalyst 1 in the presence of NBS, K2CO3 and TsNH2.
- This article is part of the themed collection: Frontiers in coordination chemistry and its applications


 Please wait while we load your content...
Please wait while we load your content...