5-Methyl-2-thienylcalcium iodide†
Abstract
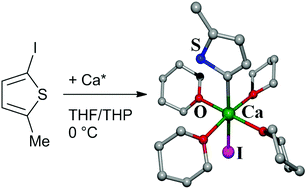
The reduction of 2-bromo- and 3-bromothiophene with calcium powder gives impure thienylcalcium complexes due to interference of various subsequent metalation and calcium–halogen exchange reactions as well as ether degradation. Therefore, calcium–iodine exchange succeeds via the reaction of trimethylsilylmethylcalcium halide with 3-iodothiophene in tetrahydrofuran at −78 °C in the presence of chloro-trimethylsilane, leading to the quantitative formation of 3-(trimethylsilyl)thiophene. The synthesis and isolation of a thienylcalcium halide, a heavy Grignard reagent with a thienyl group, is possible via the reduction of 2-iodo-5-methylthiophene with activated calcium in THF. Substitution of the thf ligands in 5-methyl-2-thienylcalcium iodide ([5-MeThien-2-Ca(thf)4I], 1) by dissolution in tetrahydropyran (THP) leads to the thp complex of 5-methyl-2-thienylcalcium iodide ([5-MeThien-2-Ca(thp)4I], 2). A low field shift of the calcium-bound carbon atom is characteristic of thienylcalcium complexes.
- This article is part of the themed collection: Organometallic and coordination chemistry of the s-block metals


 Please wait while we load your content...
Please wait while we load your content...