Crystalline protein scaffolds as a defined environment for the synthesis of bioinorganic materials
Abstract
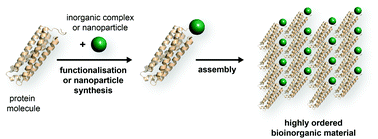
Proteins are a powerful scaffold for the organisation of inorganic components in the solid state. Due to the defined atomic structure of the protein scaffold, organometallic complexes and nanoparticles can be arranged with high precision. Moreover, crystalline protein materials are porous and enable confinement of inorganic components on the nanoscale. In this Frontier article, we summarize current strategies for the synthesis of bioinorganic materials and highlight recent examples for application of these materials. Future perspectives are discussed such as tunability of material properties through protein design, and the possibility to produce protein materials in a sustainable manner.
- This article is part of the themed collections: 2018 Frontier and Perspective articles and New Talent: Europe


 Please wait while we load your content...
Please wait while we load your content...