Structural diversity in pyridine and polypyridine adducts of ring slipped manganocene: correlating ligand steric bulk with quantified deviation from ideal hapticity†
Abstract
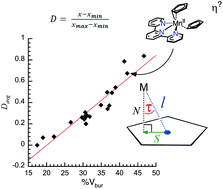
We have synthesized several new manganocene-adduct ([Cp2Mn(L)] = 1–L) complexes using pyridine and polypyridine ligands and report their molecular structures and characterization data. Consistent with other molecules in this class [(ηx-Cp)2MnLn] or [(ηx-Cp)2Mn(L–L)] (n = 1, 2; x = 1, 3, or 5), the manganese-cyclopentadienide interaction deviates from the classical ηx interactions (x = 3 or 5). Such deviations have been ascribed to steric factors and often called non-ideal hapticity. However, there is no quantification of this non-ideal hapticity and thus it is difficult to evaluate the extent of ring slippage or assign hapticity. Furthermore, the hypothesis that non-ideal hapticity in high-spin MnII complexes is induced by steric interactions has not been systematically evaluated. Therefore, we report herein a quantified scale for deviation from ideal hapticity between zero (ideal η5 interaction) and one (“η1” interaction). This quantified deviation from ideal hapticity has an empirical relationship with the ligand's steric properties, which strongly supports the premise that steric interactions cause the deviations in ionic M–Cp interactions.


 Please wait while we load your content...
Please wait while we load your content...