The first lead cobalt phosphite, PbCo2(HPO3)3†
Abstract
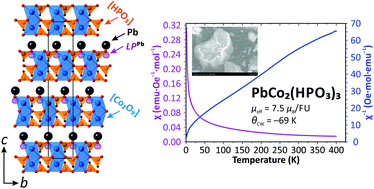
Single crystals of a new lead cobalt phosphite, PbCo2(HPO3)3, have been synthesized using mild hydrothermal techniques and characterized by X-ray diffraction analysis, SQUID magnetic measurements, IR spectroscopy, UV/vis spectroscopy, thermogravimetric analysis, and scanning electron microscopy. PbCo2(HPO3)3 crystallizes in the non-centrosymmetric (NCS) R3m space group, a = 5.3145(15) Å, c = 25.494(7) Å, V = 623.6(4) Å3. The crystal structure of PbCo2(HPO3)3 is based upon 2D heteropolyhedral blocks built up from Co2O9 octahedral dimers and HPO3 pseudo-tetrahedra. Lead cations reside in the interlayer space of the structure. Here, the NCS character results reasonably from the cooperative Pb2+ lone electron pair arrangements, by analogy to the centrosymmetric compound (NH4)2Co2(HPO3)3 with similar but disordered blocks. A local twisting of specific HPO3 groups arises due to unreasonably short H⋯H contacts between two phosphite oxoanions. In terms of the magnetic behavior, the new PbCo2(HPO3)3 phase demonstrates weak antiferromagnetic interactions inside the Co2O9 dimers between cobalt ions as expected from the phosphite μ-O bridges.


 Please wait while we load your content...
Please wait while we load your content...