Antiproliferative activity of (η6-arene)ruthenacarborane sandwich complexes against HCT116 and MCF7 cell lines†‡
Abstract
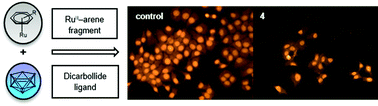
Three [(η6-arene)RuC2B9H11] complexes (arene = p-cymene (2), biphenyl (3) and 1-Me-4-COOEt-C6H4 (4)) were synthesised according to modified literature procedures and fully characterised. 2–4 were found to be moderately active against two types of tumour cell lines (HCT116 and MCF7), with IC50 values in the low micromolar range. However, viability of normal, healthy cells (MRC-5 cell line, MLEC and mouse macrophages) was not affected by treatment with 2–4, indicating high selectivity of the metallacarborane complexes towards tumour cell lines, compared to the unselective antitumour agent cisplatin and other potential RuII drugs. Moreover, flow cytometric analysis suggested that 4 induces cell death via a caspase-dependent apoptotic mechanism. DFT calculations of the frontier molecular orbitals showed that the HOMO–LUMO gap in 2–4 is smaller than in the corresponding cyclopentadienyl complexes 2-Cp–4-Cp (e.g. 5.47 (2) vs. 6.31 eV (2-Cp)). In order to assess the stability of 2–4, particularly the ruthenium–dicarbollide bond, energy decomposition analysis (EDA) of 2–4, together with the respective cyclopentadienyl analogues 2-Cp–4-Cp, was performed. EDA suggests that the ruthenium(II)–dicarbollide bond in the three complexes is mostly ionic and far stronger than the ruthenium(II)–arene bond.
- This article is part of the themed collection: Main Group Transformations

 Please wait while we load your content...
Please wait while we load your content...