pH-Dependent structural diversity of a 2-pyridinemethanol Cu complex and its relatively strong magnetic exchange coupling via hydrogen bonding†
Abstract
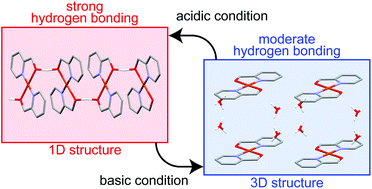
The diversity of the self-assembled structures of a Cu complex via hydrogen bonding, including a square planar unit, is reported. The Cu complex forms two self-assembled structures by hydrogen bonding, depending on the acidity of the recrystallization conditions. A linear chain structure can be produced under acidic conditions, and a three-dimensional network structure is observed under basic conditions. The linear chain structure is constructed from intermolecular sharing of a hydrogen atom between two 2-pyridinemethanolate units, with an O⋯O distance of 2.412(1) Å and an O–H–O bond angle of 170(3)°. The linear chain structure exhibits relatively strong magnetic coupling (J = −9.91(2) cm−1) via hydrogen bonding between Cu atoms; this coupling was also confirmed by electron spin resonance experiments. Thermochromic behavior was also observed for the complex.


 Please wait while we load your content...
Please wait while we load your content...