Two copper(i) cyanide coordination polymers modified by semi-rigid bis(benzimidazole) ligands: syntheses, crystal structures, and electrochemical and photocatalytic properties†
Abstract
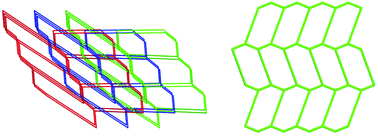
Two Cu(I) cyanide coordination polymers (CPs), namely, [Cu2(L1)(CN)2]n (1) and [Cu2(L2)(CN)2]n (2) (L1 = 4,4′-bis(2-methylbenzimidazol-1-ylmethyl)biphenyl, L2 = 4,4′-bis(5,6-dimethylbenzimidazol-1-ylmethyl)biphenyl) were synthesized and structurally characterized by single crystal X-ray diffraction, IR spectroscopy, X-ray powder diffraction and elemental analysis. The cyanide ligands in these CPs are generated in situ from the C–C bond cleavage of acetonitrile under solvothermal conditions, which is environmentally friendly and used conveniently. CP 1 features a three-fold interpenetration 3D framework consisting of Cu10(CN)6(L1)4 rings, which represents the first investigation on introducing bis(benzimidazole) ligands into copper(I) cyanide CPs with ThSi2 topology, while CP 2 exhibits a two-dimensional (6,3) layered structure containing Cu6(CN)4(L2)2 rings. The thermal stabilities, and photoluminescence and electrochemical behavior in the solid state of CPs 1 and 2 have been investigated in detail. Moreover, both CP 1 and CP 2 manifest promising photocatalytic activities (photodegradation efficiency using CP 1 is 90.8% and using CP 2 is 87.2%) for the degradation of methylene blue (MB) under UV light irradiation. A possible photocatalytic mechanism is suggested by introducing t-butyl alcohol (TBA) as a widely used ˙OH scavenger.

 Please wait while we load your content...
Please wait while we load your content...