Palladacycles of unsymmetrical (N,C−,E) (E = S/Se) pincers based on indole: their synthesis, structure and application in the catalysis of Heck coupling and allylation of aldehydes†
Abstract
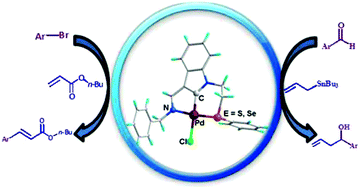
Unsymmetrical (N,C,E)-type pincer ligand precursors [L1 and L2: E = S/Se] with an indole core were synthesized for the first time by the condensation of 1-(2-phenylsulfanyl/selenylethyl)-1H-indole-3-carbaldehyde with benzyl amine. The synthetic protocols are easy and give good yields (>85%). L1 and L2 on reaction with sodium tetrachloropalladate(II) in the presence of CH3COONa result in complexes [Pd(L1/L2–H)Cl] (1/2), where they bind in a tridentate (N,C−,E) mode. L1 and L2, their aldehyde precursors and Pd(II)-complexes, 1 and 2, have been characterized by 1H, 13C{1H} and 77Se{1H} NMR and HR-MS. Palladium(II) complexes 1 and 2 and precursor aldehydes of L1 and L2 were verified using single crystal X-ray diffraction. The catalytic activities of complexes 1 and 2 were investigated for Heck coupling and allylation of aldehydes. The two reactions require 0.1–0.3 and 1 mol% loading of complexes as catalysts, respectively.

 Please wait while we load your content...
Please wait while we load your content...