Dual emission tunable in the near-infrared (NIR) and visible (VIS) spectral range by mix-LnMOF†
Abstract
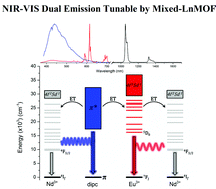
In this study, we describe the synthetic approach, crystallographic structure, luminescent behavior and elucidation of the channels of the energy conversion in heteronuclear coordination polymers with emission in the visible (Eu3+ and organic ligand) and near-infrared (Nd3+) range. The [(Nd0.9Eu0.1)2(dipc)3(H2O)3]n·nH2O, [(Nd0.7Eu0.3)2(dipc)3(H2O)3]n·nH2O, [(Nd0.5Eu0.5)2(dipc)3(H2O)3]n·nH2O, [(Nd0.3Eu0.7)2(dipc)3(H2O)3]n·nH2O, [(Nd0.1Eu0.9)2(dipc)3(H2O)3]n·nH2O, [Eu2(dipc)3(H2O)3]n·nH2O and [Nd2(dipc)3(H2O)3]n·nH2O materials are obtained by hydrothermal conditions from pyridine-2,6-dicarboxylic acid (H2dipc) and Ln2O3 oxide (Ln = Eu and Nd). The fine structures in the emission spectrum and spectral profile are used to investigate the ion responsible for the emission characteristics of a material (6), based on coordination polyhedron. The heteronuclear systems show emission in the dual spectral range (NIR-VIS) tuned for blue or red. The tuning of emission on the red (Eu3+) or blue (organic ligand) range may be performed by controlling the stoichiometric ratio of the lanthanide ions and by controlling the excitation wavelength. Nd3+ ions display self-absorption of emission to dipc ligand, resulting in interference on the emission band profile ranging from 400 to 600 nm. The energetic process of energy transfer is operated by a cascade of energy transfer, from dipc ligand mainly to Eu3+ ions and finishing on the Nd3+ ion. The efficient sensitization to Nd3+ by Eu3+ ions is due to the presence of many resonant energy levels and the short distance between these ions.

 Please wait while we load your content...
Please wait while we load your content...