Fac and mer isomers of Ru(ii) tris(pyrazolyl-pyridine) complexes as models for the vertices of coordination cages: structural characterisation and hydrogen-bonding characteristics†
Abstract
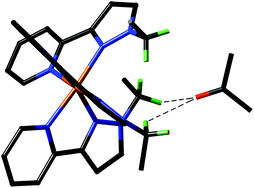
We have prepared a series of mononuclear fac and mer isomers of Ru(II) complexes containing chelating pyrazolyl-pyridine ligands, to examine their differing ability to act as hydrogen-bond donors in MeCN. This was prompted by our earlier observation that octanuclear cube-like coordination cages that contain these types of metal vertex can bind guests such as isoquinoline-N-oxide (K = 2100 M−1 in MeCN), with a significant contribution to binding being a hydrogen-bonding interaction between the electron-rich atom of the guest and a hydrogen-bond donor site on the internal surface of the cage formed by a convergent set of CH2 protons close to a 2+ metal centre. Starting with [Ru(LH)3]2+ [LH = 3-(2-pyridyl)-1H-pyrazole] the geometric isomers were separated by virtue of the fact that the fac isomer forms a Cu(I) adduct which the mer isomer does not. Alkylation of the pyrazolyl NH group with methyl iodide or benzyl bromide afforded [Ru(LMe)3]2+ and [Ru(Lbz)3]2+ respectively, each as their fac and mer isomers; all were structurally characterised. In the fac isomers the convergent group of pendant –CH2R or –CH3 protons defines a hydrogen-bond donor pocket; in the mer isomer these protons do not converge and any hydrogen-bonding involving these protons is expected to be weaker. For both [Ru(LMe)3]2+ and [Ru(Lbz)3]2+, NMR titrations with isoquinoline-N-oxide in MeCN revealed weak 1 : 1 binding (K ≈ 1 M−1) between the guest and the fac isomer of the complex that was absent with the mer isomer, confirming a difference in the hydrogen-bond donor capabilities of these complexes associated with their differing geometries. The weak binding compared to the cage however occurs because of competition from the anions, which are free to form ion-pairs with the mononuclear complex cations in a way that does not happen in the cage complexes. We conclude that (i) the presence of fac tris-chelate sites in the cage to act as hydrogen-bond donors, and (ii) exclusion of counter-ions from the central cavity leaving these hydrogen-bonding sites free to interact with guests, are both important design criteria for future coordination cage hosts.

 Please wait while we load your content...
Please wait while we load your content...