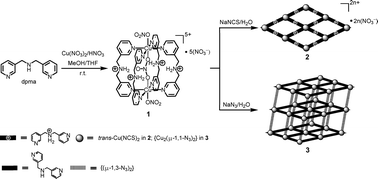
A novel M2L4 tetragonal metalloprism, [(NO3−)⊂{Cu2(μ-Hdpma)4}(NO3)2](NO3)5 (1), was prepared from the self-assembly reaction of Cu(NO3)2·3H2O and flexible clip-like organic ligand di(3-pyridylmethyl)amine (dpma) under acidic conditions. The cationic prismatic hollow structure of 1hosts one nitrate anion via both metal–ligand dative bonds and electrostatic interactions. Metalloprism 1 can dissolve in water and its prismatic structure remains intact as supported by ESI-MS data. When metalloprism 1 was treated with sodium thiocyanate and sodium azide in aqueous solutions, two polymeric coordination architectures, [Cu(μ-Hdpma)2(NCS)2](NO3)2 (2) and [Cu(μ-dpma)2(μ-1,1-N3)(μ-1,3-N3)] (3), formed at room temperature, respectively. Polymer 2 has a two-dimensional sheet structure showing a simple rhombic 44-sql topology in network connectivity, whereas polymer 3 gives a three-dimensional uninodal pcu net. The conformation of the flexible ditopic ligand is varied from a trans-trans-syn conformer in 1 to a trans-trans-anti conformer in 2 and to a trans-gauche-anti conformer in 3. The observations imply the occurrence of structural transformation from a discrete metalloprism into polymeric coordination architectures via a decoordination/rearrangement process. Magnetic studies of metalloprism 1 suggest that the two Cu(II) centers are weakly antiferromagnetically coupled. The spins communicate via the nitrate template while the Cu⋯O(nitrate) interactions are weak. For polymer 3, a ferromagnetically coupled system (J2 = +17.6 cm−1) is operative between two Cu(II) centers bridged by end-on azidos and an antiferromagnetic coupling (J1 = −7.7 cm−1) between two Cu(II) centers with end-to-end azidos. In contrast to relatively large coupling values of the reported examples, the weak ferromagnetic interaction results from insufficient spin delocalization between two Cu(II) centers.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...