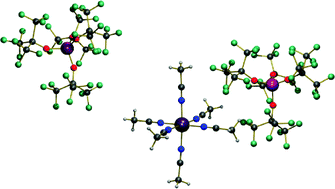
The synthesis and characterization of a series of cationic copper(II) complexes of the type [Cu(NCR)6][Al(OC(CF3)2R′)4]2 (R = CH3, Ph; R′ = CF3, Ph, PhCH3), incorporating poly/perfluoronated alkoxyaluminates as weakly coordinating anions (WCAs) is presented. Aziridination of various olefins, such as the unreactive olefinse.g.ethylhex-2-enoate and 1-decene, with N-tosyliminophenyliodinane catalyzed by [Cu(NCR)6][Al(OC(CF3)2R′)4]2 affords very good yields (up to 96%) and high TOFs (up to 5000 h−1) under mild conditions. Using disubstituted olefins as substrates, high stereoselectivities are obtained at room temperature. The to date highest cis : trans ratio (98 : 2) of the obtained aziridines is achieved for cis-stilbene in good yield (85%) as well as promising TOF (> 2000 h−1). The investigation of the solvent effect on yield and selectivity reveals that for certain oleophilic substrates (1-decene), less polar solvents, such as dichloromethane are a better choice than acetonitrile, which is commonly considered as the best solvent for olefin aziridination. Accordingly, a mechanism involving a paramagnetic copper nitrene intermediate with both concerted and stepwise pathways is proposed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...