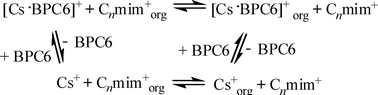
The removal of radioactive 137Cs from nuclear waste is of great importance for both the environment and energy saving. Herein, we report a study on the removal of Cs+ using a calix crown ether bis(2-propyloxy)calix[4]crown-6 (BPC6) in ionic liquids [Cnmim][NTf2], where [Cnmim]+ is 1-alkyl-3-methylimidazolium and [NTf2]− is bis(trifluoromethylsulfonyl)imide. The BPC6/[Cnmim][NTf2] system is highly efficient in removing Cs+ from aqueous solution, even at a low concentration of BPC6. HNO3 and metal ions such as Na+, Al3+ in the aqueous phase interfered with the extraction of Cs+ by competitive interaction with BPC6 and/or salting-out effect. UV analysis confirmed that the extraction of Cs+ by the BPC6/[Cnmim][NTf2] system involves a dual extraction mechanism, i.e., via exchange of BPC6·Cs+ complex or Cs+ by [Cnmim]+. Irradiation of [C4mim][NTf2] dramatically decreases Cs+ partitioning in the ionic liquid phase by the competitive interaction of radiation-generated H+ with BPC6, while irradiation of BPC6/[C4mim][NTf2] decreases Cs+ partitioning more markedly due to the radiolysis of BPC6.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...