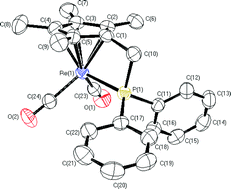
The fulvene complex (η6-C5Me4CH2)Re(C6F5)(CO)2 reacts at the exocyclic methylene carbon with potassium diphenylphosphide to yield the anionic species [(η5-C5Me4CH2PPh2)Re(C6F5)(CO)2]− (1). Protonation of 1 with HCl at 0 °C produces the hydride complex trans-(η5-C5Me4CH2PPh2)Re(C6F5)(H)(CO)2 (2). Thermolysis of a hexanes solution of 2, under nitrogen atmosphere, produces the chelated complex (η5:η1-C5Me4CH2PPh2)Re(CO)2 (3) in good yield. The thermolysis under a CO atmosphere affords a mixture of the complexes (η5:η1-C5Me4CH2PPh2)Re(CO)2 (3) and (η5-C5Me4CH2PPh2)Re(CO)3 (4a). The reaction of 3 with two electron donor ligands yields (η5-C5Me4CH2PPh2)Re(CO)2(L) (4a, L = CO; 4b, L = PMe3; 4c, L = tBuNC). Complex 3 also reacts with I2, HBF4 and MeOTf to yield the cationic compounds trans-[(η5:η1-C5Me4CH2PPh2)Re(R)(CO)2]+ (5a, R = I; 5b, R = H; 5c, R = Me). Upon treatment with chloroform, the hydride complex 5b converts to the corresponding chloro derivative 5d. The trans stereochemistry for complexes 5 have been assigned on basis of ν(CO) IR intensities and 13C-NMR chemical shifts. The reaction of the cationic complexes (5a, 5c) with KI and Me3NO·2H2O yields the neutral species cis-(η5:η1-C5Me4CH2PPh2)Re(I)(R)(CO) (6a, R = I, 6b, R = Me). The molecular structure of 3 and 6a have been determined by X-ray crystallography.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...