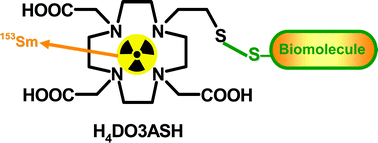
The novel tetraazamacrocycle 10-(2-sulfanylethyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triacetic acid (H4DO3ASH) was synthesized and characterized by multinuclear NMR spectroscopy, 2D NMR techniques and mass spectrometry. The protonation constants of H4DO3ASH were determined by potentiometry at 25 °C in 0.1 M KCl ionic strength, and the protonation sequence was assigned based on 1H- and 13C-NMR titrations. The stability constants of the DO3ASH complexes with Ce3+, Sm3+ and Ho3+ have been determined by potentiometry and UV-Vis spectroscopy. They are very similar, comprising a narrow range (logKML = 21.0–22.0). UV-Vis spectrophotometric data on Ce3+–DO3ASH and relaxivity measurements on the Gd3+–DO3ASH complex suggest that the thiol group does not coordinate to the metal, even in its deprotonated form. For labeling with radioactive lanthanides(III), various conditions were tested and both complexes, 153Sm/166Ho–DO3ASH, were obtained in quantitative yield (> 98%) at pH = 6. At room temperature, formation kinetics were faster for the 153Sm than for the 166Ho complex (5 vs. 60 min, respectively, needed for complete labeling). The stability of these hydrophilic complexes (153Sm, logD = −2.1; 166Ho, logD = −1.6) has been studied in different buffers, in human serum and in the presence of excess of cysteine and glutathione. 153Sm–DO3ASH has shown a high stability under these conditions and a relatively low protein binding (2.1%), while 166Ho–DO3ASH was less stable, including in the presence of cysteine and glutathione, and had a slightly higher protein binding (6.7%). In vivo studies have been performed only for the more stable 153Sm–DO3ASH complex and its biological profile and in vivo stability has been compared to that of 153Sm–DO3A in the same animal model. The biodistribution profile presents a similar trend with rapid total excretion from the whole animal body, mainly via the urinary pathway. The most striking difference found is related to a slightly slower clearance of 153Sm–DO3ASH from organs like blood, bone and muscle as compared to 153Sm–DO3A. Additionally, the fraction of 153Sm–DO3ASH taken by the hepatobiliar tract is also modestly higher than that of 153Sm–DO3A.

 Please wait while we load your content...
Please wait while we load your content...