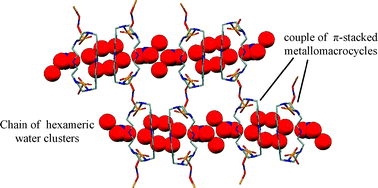
The synthesis and characterisation of a new macrocyclic compound (L), composed of a pentamine chain linking the 2,7 positions of an acridine moiety, is reported. Cu(II) complexation was studied by means of potentiometric, UV-vis and EPR measurements in aqueous solutions. This study reveals that the ligand forms a stable tetranuclear complex with an overall [Cu4L2(OH)4]4+ stoichiometry in aqueous solution. The crystal structure of the [Cu8L4(μ-OH)8(μ-NO3)3](NO3)5·32H2O complex, isolated from neutral aqueous solution, shows that it is formed by tetranuclear clusters [Cu4L2(μ-OH)4(μ-NO3)x](4−x)+ (x= 1 or 2), composed of two [Cu2L]4+ macrocyclic units linked together by four bridging hydroxide and nitrate anions to give an overall metallomacrocyclic structure. Magnetic measurements shows that the hydroxide-bridged Cu(II) ions are ferromagnatically coupled. Inspection of the crystal packing shows that couples of metallomacrocycles are paired by π-stacking interactions between acridine moieties, giving rise to an internal hydrophilic cavity were hexameric or pentameric water clusters are enclosed. The coupled metallomacrocycles assume a columnar disposition growing along the a axis, giving rise to channels passing through the cavities of the metallomacrocycles; these channels are filled by chains of 5- or 6-membered water clusters linked together by [NO3·H2O]2 units.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...