New synthesis of zinc tetrakis(arylethynyl)porphyrins and substituent effects on their redox chemistry
Abstract
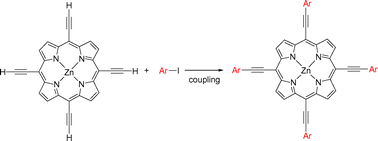
Sonogashira coupling of zinc 5,10,15,20-tetraethynylporphyrin with various phenyl iodides under mild conditions afforded good yields of the corresponding zinc porphyrins. This method is applicable to a variety of aryl iodides including meso-substituted iodoporphyrin to form a conjugated star-shaped multiporphyrin. The UV-Vis spectra show that peak broadening, red shifts, and changes in the oscillator strength of absorptions increase with the extension of π-conjugation. In the electrochemical measurements, the first oxidation of porphyrins 4–9 occurs at potentials in the range +0.89 to +1.08 V, which are comparable to that of ZnTPP (TPP = tetraphenylporphyrin). The first reduction was observed at potentials from −0.73 to −0.89 V, which is anodically shifted by 390–550 mV as compared to that of ZnTPP, and the second reduction occurs at potentials in the range −1.12 to −1.33 V. The para-substituted tetrakis(phenylethynyl)porphyrins show substituent effects on their redox chemistry and exhibit only slight substituent effects in their emission and absorption maxima.

 Please wait while we load your content...
Please wait while we load your content...