A facile synthetic access to the homologous series of 1,1′-ferrocenylene bisdihalophosphanes Fc′(PX2)2 (X = F, Cl, Br, I; Fc′ = 1,1′-ferrocenediyl = ferrocenylene = –C5H4FeC5H4–) is reported. The 31P NMR spectroscopic data of this series suggest a similar electronic interaction of the –PX2 unit with the ferrocene system as in monofunctional ferrocenyldihalophosphanes. Crystal structures for Fc′(PCl2)2 and Fc′(PBr2)2 reveal that the nature of the halogen atom within the phosphane unit strongly influences the twist angle of the ferrocene system, while leaving the other structural parameters mostly unaffected. Based on these bisdihalophosphanes, a synthetic access to the first metallocene bridged bisphosphaalkene ((C5H4P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(t-Bu)OTMS)2Fe) is reported in which the tert-butyl substituents provide sufficient steric pressure to control the E/Z isomeric ratio which leads to the almost exclusive formation of the most stable Z,Z isomer out of the three possible isomers.
C(t-Bu)OTMS)2Fe) is reported in which the tert-butyl substituents provide sufficient steric pressure to control the E/Z isomeric ratio which leads to the almost exclusive formation of the most stable Z,Z isomer out of the three possible isomers.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(t-Bu)OTMS)2Fe) is reported in which the
C(t-Bu)OTMS)2Fe) is reported in which the 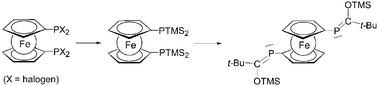

 Please wait while we load your content...
Please wait while we load your content...