Photochemical and thermal synthesis and characterization of polypyridine ruthenium(ii) complexes containing different monodentate ligands†
Abstract
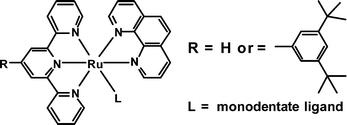
The photochemical and thermal synthesis of a series of [Ru(tpy)(phen)L]2+ complexes where terpy = 2,2′:6′,2″–terpyridine (terpy) or 4-(3,5-di-tert-butyl)phenyl-2,2′;6′,2″-terpyridine (terpy*), phen = 1,10-phenanthroline and L monodentate ligands such as Cl−, NC−, CH3CN, pyridine, isoquinoline (iq), 4-dimethylaminopyridine (dmap), 4-(4′-methylpyridinium)pyridine (mqt), phenothiazine (ptz), 3,5-lutidine(lut) and H2O are described. The complexes have been characterized by 1H (COSY and ROESY) NMR spectroscopy, mass spectrometry (FAB and ES-MS), cyclic voltammetry, UV-vis absorption and emission spectroscopy and single-crystal X-ray diffraction. Photochemical experiments have shown that the ligands L can be photochemically expelled and replaced by CH3CN molecules used as solvent. This type of ligand interchange occurs efficiently and very selectively. The reaction has the potential to be applied to a wide range of entering ligands (thioethers, ethers, pyridines, sulfoxides, nitriles, amides) and indicates the high stability of the Ru(terpy)(phen) core under the irradiation conditions.

 Please wait while we load your content...
Please wait while we load your content...