The diphosphonic acid, N,N′-dimethyl-N,N′-ethylenediamine-bis(methylenephosphonic acid)
(H4L)
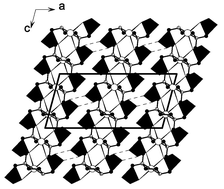
1, along with its Ni(II) and Pb(II) complexes, have been synthesized and structurally determined. The structure of the diphosphonic acid features a 2D layer built from the molecular units interconnected by hydrogen bonds between phosphonate oxygen atoms and/or nitrogen atoms. Upon complexing with the Ni(II) ion, the diphosphonic acid acts as a tetradentate chelating ligand (2N, 2O), forming Ni(H2L)(H2O)22 molecular units, such units are also interconnected into a hydrogen bonded double layer via hydrogen bonds between uncoordinated phosphonate oxygen atoms. The ligand in Pb(H2L)
3 also chelates with a Pb(II) ion tetradentately, however, neighboring Pb(H2L) units are interconnected into a one-dimensional chain along the c-axis by a pair of μ2-chelating and bridging phosphonate oxygen atoms. Such chains are further interconnected into a 3D network via hydrogen bonds between uncoordinated phosphonate oxygen atoms. These structures demonstrate the important role of hydrogen bonds in the chemistry of divalent metal phosphonates.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...