Central vs. peripheral Ag(i) coordination in NS3-open chain and cage ligands
Abstract
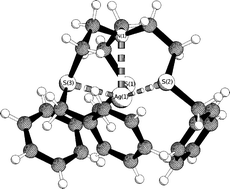
A series of NS3-open chain and cage ligands were synthesised and their complexation behaviour towards Ag(I) and Cu(I) studied. Crystal structures show that all open chain ligands form complexes in which the four donor atoms of the ligands coordinate the metal ions in a trigonal pyramidal geometry. For the NS3-cages, however, the ions are not in the centre of the cage, but show peripheral coordination resulting in polymeric crystal structures. The new NS3-aromatic cage ligand 18 binds Ag(I) peripherally giving a polymeric structure in the solid state and fluxional behaviour in solution. NMR evidence for equally populated central and peripheral coordination sites is coherent with results from DFT calculations.

 Please wait while we load your content...
Please wait while we load your content...