Synthesis and structural characterization of Group 4 metallocene complexes that contain remote carboxamide functionalities at their Cp-side chains†
Abstract
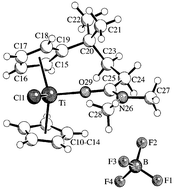
Treatment of the levulinic acid amides CH3COCH2CH2CONR27 [–NR2 = NMe2 (a), NEt2 (b), pyrrolidino (c)] with cyclopentadiene in the presence of excess pyrrolidine gave the corresponding pentafulvene derivatives 8(a–c). Their subsequent reaction with [Me2CuLi] followed by hydrolysis furnished the corresponding C-methylated cyclopentadienes 9(a–c), each obtained as a mixture of isomers (9-A and 9-B). Deprotonation of the cyclopentadiene units in these compounds was achieved by treatment with lithium diisopropylamide (LDA) to yield the substituted lithium cyclopentadienide reagents [(C5H4)–CMe2CH2CH2CONR2]Li (10a–c), that were resistant to intramolecular cyclization under the applied reaction conditions. The transmetalation reaction of 10a with FeCl2 furnished the ferrocene derivative [(C5H4)–CMe2CH2CH2CONMe2]2Fe (11a). Treatment of 10a–c with CpTiCl3 gave the Group 4 bent metallocene complexes Cp[(C5H4)–CMe2CH2CH2CONR2]TiCl2 (12a–c). Similarly, the reaction of 10b with CpZrCl3 led to formation of the complex Cp[(C5H4)CMe2CH2CH2CONEt2]ZrCl2 (13b). Treatment of 12a with Meerwein's reagent [(Et3O+)(BF4−)] resulted in chloride abstraction with formation of the metallocene cation complex 16 (with BF4− anion), in which, contrary to the 16-electron systems 12 and 13, the carboxamido oxygen atom intramolecularly coordinates to the electron deficient Group 4 metal cation. The organic reagents 8a and 9a as well as the transition metal complexes 11a, 13b, and 16[BF4−] were characterized by X-ray crystal structure analyses.

 Please wait while we load your content...
Please wait while we load your content...