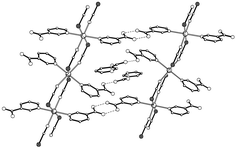
Four novel non-interpenetrating metal–organic co-ordination networks of the CdII–L–NCS− system have been prepared using potentially bridging nicotinic acid derivatives L [nicotinic acid (HL1), nicotinamide (L2), isonicotinamide (L3), or isonicotinate (L4)]. Their crystal structures were determined by X-ray diffraction. In [Cd(SCN)2(HL1)2]·HL11 each pair of cadmium(II) ions is bridged by two inversely related μ-NCS−-N,S ligands to form infinite chains with the remaining two trans positions of six-co-ordinated Cd atoms being occupied by two HL1 ligands, which form head-to-head double hydrogen bonds using the unco-ordinated carboxyl groups between adjacent chains to form two-dimensional layers. Weak S ⋯ S interactions between the NCS− ligands extend the layers into a three-dimensional framework with the channels enclosing HL1 guest molecules, which are interlinked into chains through O–H ⋯ N hydrogen bonds. [Cd(SCN)2(L2)2]·H2O 2 and [Cd(SCN)2(L3)2] 3 are interesting in that they contain unprecedented 16-membered [Cd4(μ-SCN-N,S )4] rings in the two-dimensional sheets. Between the sheets N–H ⋯ O amide–amide hydrogen bonds extend the two-dimensional layers to three-dimensional structures. [Cd(SCN)(L4)(H2O)] 4 exhibits two-dimensional wave-like networks with bridging NCS− and L4 ligands using both ends in co-ordination. The results demonstrate that the structures of the CdII–L–NCS system deeply depend on the nature of the nicotinic acid derivatives L.
)4] rings in the two-dimensional sheets. Between the sheets N–H ⋯ O amide–amide hydrogen bonds extend the two-dimensional layers to three-dimensional structures. [Cd(SCN)(L4)(H2O)] 4 exhibits two-dimensional wave-like networks with bridging NCS− and L4 ligands using both ends in co-ordination. The results demonstrate that the structures of the CdII–L–NCS system deeply depend on the nature of the nicotinic acid derivatives L.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 )4] rings in the two-dimensional sheets. Between the sheets N–H ⋯ O amide–
)4] rings in the two-dimensional sheets. Between the sheets N–H ⋯ O amide–

 Please wait while we load your content...
Please wait while we load your content...