Selective oxidation of methane to methanol on dispersed copper on alumina from readily available copper(ii) formate†
Abstract
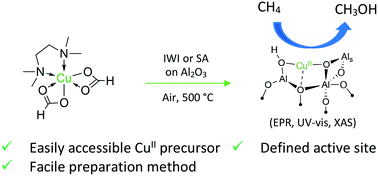
The direct conversion of methane to methanol attracts increasing interest due to the availability of low-cost methane from natural gas. Significant steps in that direction have already been achieved, using a stepwise chemical looping approach employing zeolites, but these materials have limited industrial relevance, because of low methanol productivity and relatively high materials cost. We therefore investigated the reactivity of copper supported on γ-Al2O3 prepared by common synthetic routes, namely incipient wetness impregnation and specific adsorption. All materials showed reactivity for the partial oxidation of methane under mild conditions (6 bar, 200 °C), with increasing reactivity as the copper loading increases, leading to a maximum methanol yield of 15 μmol CH3OH g−1. A combination of spectroscopic techniques (UV-vis, XAS, EPR, TPR) allowed the assignment of the reactive species at low loading to the previously reported monomeric [CuO(OH)]− sites. This straightforward synthetic approach and spectroscopic characterization of the Cu(II) speciation can be applied to a wide range of oxide supports.


 Please wait while we load your content...
Please wait while we load your content...