Sulfur-doped g-C3N4 for efficient photocatalytic CO2 reduction: insights by experiment and first-principles calculations†
Abstract
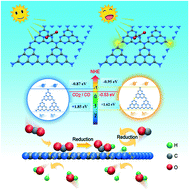
The development of doped g-C3N4 photocatalyst has demonstrated potential advantages for the artificial photosynthesis of hydrocarbon fuels by utilizing solar energy and CO2. Herein, a new two-dimensional S-doped g-C3N4 (S-CN) was designed and synthesized. S-CN displayed a high CO evolution rate of 16.02 μmol g−1 in water (10 times more than bulk g-C3N4) at a molar ratio of thiourea to DCNA of 0.4. Also, S-CN could maintain its activity for up to 15 h during the stability test. S–C bond formation was revealed for the first time by combining material characterization and density functional theory (DFT) calculations. The S 3p state made an excellent contribution to moving up the conduction band position and altering the S-CN band gap. The doped S atoms caused charge rearrangement and significantly enhanced the electron–hole separation, enhancing the CO2 reduction activity compared to bulk-CN. The present work provides a broadening window for the development of non-metal-doped g-C3N4 with outstanding CO2 photoreduction performance.


 Please wait while we load your content...
Please wait while we load your content...