Self-assembly of a highly stable and active Co3O4/H-TiO2 bulk heterojunction with high-energy interfacial structures for low temperature CO catalytic oxidation†
Abstract
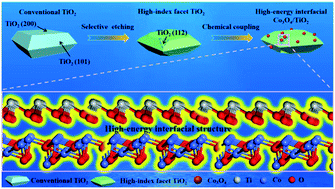
Development of transition metal oxide catalysts with a tunable structure is extremely important for energy, chemical and environmental processes. Herein, we report a new strategy for the synthesis of a highly stable and active Co3O4/H-TiO2 bulk heterojunction with high-energy interfacial structures through two-step hydrothermal-crystallization for efficient low-temperature CO catalytic oxidation. By selectively etching, an anatase TiO2 support exposing the high-index (112) facet was first created. The high-index (112) facet with high surface energy can endow the bulk TiO2 support with unique physical and chemical properties so as to act as a foundation to construct a robust high-energy interfacial bulk heterojunction. Because of the presence of Ti–O–Co species at the high-energy interface, the prepared Co3O4/H-TiO2 bulk heterojunction possessed much more catalytic active sites compared to Co3O4 nanoparticles supported on conventional TiO2 supports and exhibited a turnover frequency (TOF) value of 3.56 × 10−3 s−1 at 120 °C, which was very competitive compared to those of reported cobalt-based catalysts and 1000 times higher than that of commercial Co3O4. Importantly, due to the strong interactions between Co3O4 and the TiO2 bulk support exposed high-index facet, more attractive water-resisting properties for low temperature CO oxidation can be achieved in the presence of steam (∼2%) compared to previously reported catalysts. The results show promising opportunities for tailoring catalytic behaviors beyond a facile and rational design with a controllable metal–support interface.


 Please wait while we load your content...
Please wait while we load your content...