Mechanistic insights into the oxidation of copper(i) species during NH3-SCR over Cu-CHA zeolites: a DFT study†
Abstract
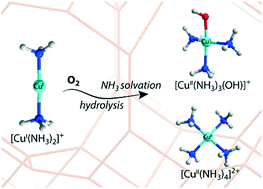
Selective catalytic reduction of nitrogen oxides using ammonia (NH3-SCR) over Cu-exchanged zeolites proceeds via reduction of Cu(II) to Cu(I) and subsequent reoxidation of Cu(I) to Cu(II). Although the mechanism of reduction half cycle has been relatively well established, reoxidation pathways of Cu(I) to form the original Cu(II) species are highly complicated and remain unclear. Herein, oxidation mechanisms of Cu(I) to Cu(II) species in CHA zeolites during the NH3-SCR process were investigated by periodic DFT calculations. The NH3-solvated Cu(I) and Cu(II) species were considered for exploring the oxidative activation reaction pathways. The results show that, with O2 as the sole oxidant, Cu(I) can be effectively oxidized to Cu(II) via multinuclear Cu-oxo intermediates with moderate reaction barriers. The NO-assisted oxidation of Cu(I) was found to favor the formation of Cu nitrate/nitrite species, which seem to only act as off-cycle resting states. We propose that reoxidation of Cu(I) to Cu(II) with O2 as the sole oxidant plays a key role in the oxidation half cycle under standard NH3-SCR conditions.
- This article is part of the themed collection: Catalysis Science & Technology 10th Anniversary Symposium


 Please wait while we load your content...
Please wait while we load your content...
