Highly tunable periodic imidazole-based mesoporous polymers as cooperative catalysts for efficient carbon dioxide fixation†
Abstract
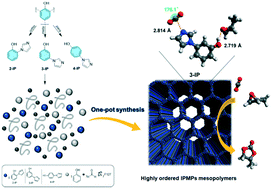
The insertion of carbon dioxide (CO2) into epoxides is a “greener” route for producing cyclic carbonates. Here we report a one-pot synthesis of highly ordered imidazolyl-functionalized mesoporous phenolic resins (IPMPs). We show that the ordering of the IPMP mesostructure depends significantly on the imidazolyl-based precursors. 3-(Imidazole-1-yl)phenol (3-IP) with unoccupied ortho and para sites was found to be the optimal precursor, yielding a highly ordered cross-linked mesopolymer (3-IPMP) with a high surface area, low density, uniform mesopores and controllable imidazole loading. Control experiments and DFT calculations show that the imidazole doped into the mesoporous organic framework can act synergistically with abundant phenolic hydroxyl groups for co-activation of CO2 and epoxides, which outperforms the classical periodic mesosilica and polymer-based catalysts. Building on these results, the 3-IPMP mesopolymer was further functionalized with various alkyl halides, forming robust ionic polymers, avoiding the eco-unfriendly addition of KI. In comparison to the porous imidazolium-based poly(ionic liquid), 3-IPMP-EtI, combines the advantages of the imidazole active sites and homogenous KI additive as well as abundant phenolic OH groups in the mesoporous hydrophobic framework, synergistically enhancing its catalytic activity in cycloaddition reactions of CO2 without the use of any co-catalyst. This novel catalyst is stable and can be reused at least five times without losing activity.


 Please wait while we load your content...
Please wait while we load your content...