Controlling the redox properties of nickel in NiO/ZrO2 catalysts synthesized by sol–gel
Abstract
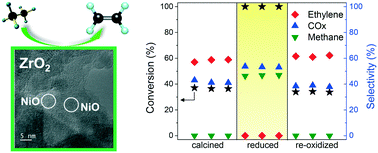
NiO–ZrO2 samples were prepared by the sol–gel method adjusting the nickel content to 3 and 10 wt% and varying the calcination temperature from 500 to 700 °C. The solids were characterized by X-ray diffraction, ultraviolet-visible diffuse reflectance spectroscopy (UV-vis DRS), X-ray photoelectron spectroscopy, high resolution transmission electron microscopy (HRTEM) and scanning and transmission electron microscopy (STEM). For the sample with 3 wt% Ni, diffraction lines related to cubic zirconia were only detected when calcined from 500 to 650 °C, a NiO crystalline phase as well as the transition phase from cubic to tetragonal zirconia appeared when calcined at 700 °C. UV-vis DRS and HRTEM results indicated the presence of a highly dispersed NiO phase at the nanometric scale throughout the main zirconia crystalline phase. The NiO crystalline phase was already detected for the sample with 10 wt% nickel content calcined at 500 °C. The NiO–ZrO2 interaction was modified by reducing the hydrolysis rate during synthesis leading to a sample with a high dispersion of Ni throughout ZrO2, thus modifying the reducibility of NiO. The NiO–ZrO2 samples were catalytically tested for the oxidative dehydrogenation of ethane as a model reaction. Prior to reaction, the calcined catalysts were pre-treated in situ under reducing and oxidant atmospheres to study their redox properties. As the NiO–ZrO2 interaction modifies the electronic properties of both nickel oxide and zirconia, ethane conversion and ethylene selectivity were strongly influenced not only by the nickel content and calcination temperature but also by the in situ pre-treatment before reaction. This effect was particularly evident in the sample prepared with a modified hydrolysis rate, which changed the redox properties of the NiO species.


 Please wait while we load your content...
Please wait while we load your content...