Improved hydrothermal stability of Pd nanoparticles on nitrogen-doped carbon supports†
Abstract
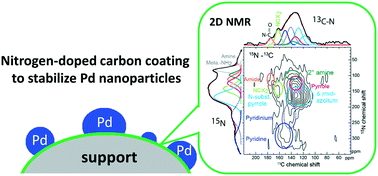
Carbon supports have been shown to provide better hydrothermal stability than alumina or silica supports, thus attracting more attention for aqueous-phase biomass conversion reactions. However, sintering and leaching of the metal particles still occur during condensed phase utilization of carbon supported metal catalysts. To further improve the stability of supported metal particles, a simple nitrogen-doped carbon coated SBA-15 was synthesized and systematically characterized. Better stability of supported Pd particles was found from nitrogen incorporation into the carbon support during extended hydrothermal treatment or under continuous flow reaction conditions. With these materials, leaching was negligible and sintering was suppressed. Based on solid-state 15N and 13C NMR analysis before and after hydrothermal treatment, the improved dispersion and stability from nitrogen doping was ascribed to pyridine, pyrrole, and imidazolium groups. Relatively small carbon fragments resulting from nitrogen incorporation rendered more silica exposure to water and concomitant hydrolysis, but the stability and activity of Pd particles were preserved. The Pd stability advantage was also observed on nitrogen-doped carbon coatings of silica gel and CMK-3 supports. The enhanced stability of Pd particles on different nitrogen-doped carbon materials was correlated with the synergistic effect of oxygen and nitrogen heteroatoms and the formation of a decorative carbon overlayer on the Pd particles.


 Please wait while we load your content...
Please wait while we load your content...
