Insights into mechanism and selectivity in ruthenium(ii)-catalysed ortho-arylation reactions directed by Lewis basic groups†
Abstract
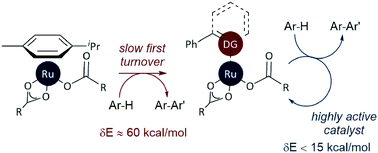
We report a detailed study of the selectivity of ruthenium-catalysed C–H arylation reactions directed by Lewis basic heterocycles. A reactivity scale for directing power in these reactions, based on the results of intermolecular competition experiments, is reported for the first time. Our work is supported by detailed density functional theory calculations that reveal the underlying mechanism of this reaction, which requires the dissociation of a p-cymene ligand before oxidative addition becomes competent. The calculated energetic span of the catalytic cycles for each substrate is broadly in agreement with our experimental observations. This work advances our understanding of mechanism and selectivity in these reactions, and provides a basis for future catalyst design efforts.


 Please wait while we load your content...
Please wait while we load your content...