Ultrasmall Ni nanoparticles embedded in Zr-based MOFs provide high selectivity for CO2 hydrogenation to methane at low temperatures†
Abstract
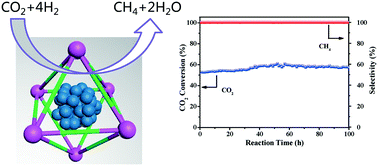
Of great significance from an energy-saving viewpoint is the direct use of CO2 as a C1 source to mitigate the anthropogenic CO2 emission into the earth's atmosphere and to produce methane that can be turned into chemicals and fuels. Herein, we report the use of UiO-66 metal–organic frameworks to anchor ultrasmall Ni nanoparticles (NPs), thus avoiding the sintering of Ni NPs protected by the frameworks. Transmission electron microscope images and EDX mappings show that Ni NPs with an average size of 2 nm are highly dispersed in UiO-66 (Ni@UiO-66). Moreover, the obtained catalyst with an optimal Ni loading of 20 wt% displays an outstanding activity (57.6% of CO2 conversion) and high selectivity (100%) for methane in long-term stability tests up to 100 h at a reaction temperature as low as 300 °C. Compared with Ni/ZrO2 and Ni/SiO2, the good dispersion of ultrasmall Ni NPs and the low activation energy (Ea = 68.9 kJ mol−1) facilitate a high catalytic activity, which makes Ni@UiO-66 a promising catalyst for CO2 methanation.


 Please wait while we load your content...
Please wait while we load your content...