Ethane dehydrogenation on pristine and AlOx decorated Pt stepped surfaces†
Abstract
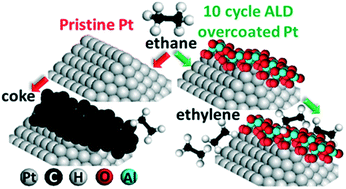
Ethane dehydrogenation on pristine and AlOx decorated Pt surfaces was studied using density functional theory (DFT) calculations and reaction kinetics experiments. Following the study of adsorption of fourteen distinct C2Hx and CHx species, the whole reaction network for ethane dehydrogenation on the terrace and the step of the pristine Pt(433) surface was systematically analyzed using DFT. We show that the under-coordinated step edge sites bind C2Hx and CHx species more strongly than terrace sites. These under-coordinated step sites facilitate deep dehydrogenation, resulting in accumulation of coke precursors on the surface and catalyst deactivation. We demonstrate that by decorating the step with AlOx species the step edge sites can be blocked, leading to substantial suppression of deep dehydrogenation and coke formation. These findings were corroborated by reaction kinetics experiments where a four-fold enhancement of the turnover frequency for ethylene formation was measured on 10-cycle atomic layer deposited (ALD) alumina overcoated Pt/γ-Al2O3 catalyst compared to Pt/γ-Al2O3 without decoration with ALD-alumina. Our results suggest that AlOx decorated Pt is a promising catalyst for alkane dehydrogenation and could have applications, including in endothermic cooling in hypersonic flights.


 Please wait while we load your content...
Please wait while we load your content...
