Metallic 1T-LixMoS2 co-catalyst enhanced photocatalytic hydrogen evolution over ZnIn2S4 floriated microspheres under visible light irradiation
Abstract
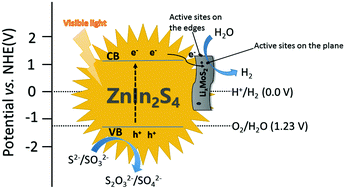
Lithium intercalated 1T allotropes of molybdenum disulfide/zinc indium sulfide (1T-LixMoS2/ZnIn2S4) composites were synthesized using a two-step hydrothermal method. The as-prepared samples were characterized using X-ray diffraction, scanning electron microscopy, transmission electron microscopy, X-ray photoelectron spectroscopy, ultraviolet-visible diffuse reflectance absorption spectra, photoluminescence emission spectra and Raman spectra. The results indicate that the lithium intercalation reaction transforms the MoS2 crystal from the hexagonal structure (2H phase) to the trigonal 1T phase. By introducing metallic 1T-LixMoS2 as a co-catalyst, the photocatalytic hydrogen (H2) evolution activity of the ZnIn2S4 microspheres is significantly enhanced under visible light irradiation. The enhanced photocatalytic activity is mainly attributed to the fact that the metallic 1T-LixMoS2 promotes the efficient separation of the photoexcited electrons and the holes of ZnIn2S4 microspheres because of its high electrical conductivity and high densities of the active sites. The photoexcited electrons of ZnIn2S4 microspheres can not only transfer to the edge sites of 1T-LixMoS2, but also to the basal plane active sites of 1T-LixMoS2. Furthermore, the photocatalytic activity is dependent on the content of 1T-LixMoS2 in the composites. The 2.0 wt% 1T-LixMoS2/ZnIn2S4 sample shows the highest H2 production rate of 332.4 μmol h−1, which is 2.4 times higher than that of bare ZnIn2S4 and 1.3 times higher than that of 2.0 wt% 2H-MoS2/ZnIn2S4, and it remains stable even after three photocatalytic cycling tests.


 Please wait while we load your content...
Please wait while we load your content...