Construction of solid–liquid interfacial Fenton-like reaction under visible light irradiation over etched CoxFeyO4–BiOBr photocatalysts†
Abstract
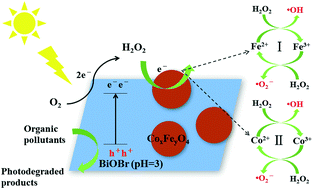
In this study, we constructed an in situ Fenton-like photocatalytic system driven by visible-light irradiation. First, BiOBr, a benign in situ H2O2 producer, was prepared via a simple hydrothermal method. Then, the accelerant (CoFe2O4 nanoparticles as the precursor) for H2O2 decomposition was loaded onto the surface of the BiOBr photocatalyst to build a well-contacted interface for reactive sites. CoxFeyO4 nanoparticles etched to different extents were obtained by adjusting the solution pH (pH = 1, 3 and 7) during the composite preparation process. A group of green and efficient CoxFeyO4–BiOBr photocatalysts (abbreviated as CFB) (pH = 1, 3 and 7) were successfully synthesized. The optimal CFB material used for BPA photodegradation was 0.5% CFB (pH = 3). The reaction rate constant of 0.5% CFB (pH = 3) was 3.4 times higher than BiOBr (pH = 3). H2O2 detection, ESR and radical trapping experiments demonstrated that much of the H2O2 produced by BiOBr (pH = 3) was successfully decomposed to ˙O2− and ˙OH, which played important roles with h+ in the BPA photodegradation process, thereby a possible photocatalytic mechanism was proposed. Cycling experiments indicated the good stability of 0.5% CFB (pH = 3). This work may offer a green and efficient method for in situ H2O2 generation and decomposition, thus, the secondary pollution and harsh conditions of the conventional Fenton reaction can be tactfully avoided.


 Please wait while we load your content...
Please wait while we load your content...