Performance of edges on carbon for the catalytic hydroxylation of benzene to phenol†
Abstract
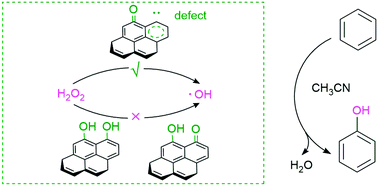
Carbon catalyst is regarded as an efficient and environment-friendly material for the direct oxidation of benzene to phenol. However, the essence and detailed performances of the active sites are still unclear at the molecular level. In the present study, the role of typical defect sites on carbon for benzene hydroxylation to phenol with hydrogen peroxide as the oxidant was theoretically investigated at M06-2x/6-311++G(d, p) level in acetonitrile solution. The carbon with defect sites was modeled with armchair and zigzag edges. The turnover frequency analysis indicated that the rate-controlling step was associated with the HO–OH bond cleavage in the H2O2 moiety for the formation of hydroxyl radical. For both the armchair and zigzag edges on carbon material, the defect site plays an essential role in the hydroxyl radical formation from H2O2. In the presence of water, the defect site of carbon could readily be hydrated, where the zigzag defect site could be more easily hydrated to hydroquinone than the defect armchair site. Furthermore, the zigzag defect carbon exhibits better catalytic performance than the armchair defect carbon. Based on activation strain analysis, there is a stronger stabilizing transition state interaction in the zigzag defect carbon than that in the defect armchair carbon, which arises from a narrower HOMO–LUMO gap. The present study could shed some light on the essence and function of active sites on carbon for the benzene hydroxylation to phenol.
- This article is part of the themed collection: 2018 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...