Sub-nano MgF2 embedded in carbon nanofibers and electrospun MgF2 nanofibers by one-step electrospinning as highly efficient catalysts for 1,1,1-trifluoroethane dehydrofluorination†
Abstract
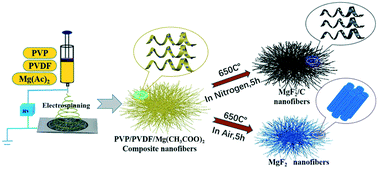
Hydrofluorocarbons (HFCs) which are usually potent greenhouse gases are regulated by the Montreal Protocol and its amendments, especially the recent Kigali Amendment. Dehydrofluorination of HFCs is an efficient route for the conversion of these greenhouse gases to value added and environmentally benign chemicals. Although AlF3 with strong Lewis acidity catalyzes dehydrofluorination, it also favors carbon deposition. MgF2 with weak acidity inhibits coking significantly. Unfortunately, MgF2 sinters dramatically at temperatures below 300 °C leading to the low activity for dehydrofluorination. In the present work, we report that sub-nano MgF2 embedded in carbon fibers and electrospun MgF2 fibers prevent sintering of MgF2 during dehydrofluorination reaction. Via simple and one-step electrospinning and calcination in a N2 (for embedded MgF2) or air (for MgF2 fibers) atmosphere, embedded MgF2 with particle sizes between 3–6 nm and pure MgF2 fibers with diameters of around 100 nm were fabricated. No sintering was observed following reaction at 450 °C. The fine MgF2 particles and MgF2 fibers facilitate the formation of under coordinated Mg species in MgF2 which are the weak acid sites. By embedding MgF2 or fabrication of MgF2 fibers, weak acid sites are increased significantly, while strong acid sites remain almost unchanged. Hence, they show significantly higher reaction rates than MgF2 prepared by precipitation of Mg(CH3COO)2·4H2O with NH4F for the dehydrofluorination of 1,1,1-trifluoroethane (HFC-143a) to VDF (CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CF2) at 450 °C.
CF2) at 450 °C.


 Please wait while we load your content...
Please wait while we load your content...