Non-oxidative dehydrogenation of propane, n-butane, and isobutane over bulk ZrO2-based catalysts: effect of dopant on the active site and pathways of product formation†
Abstract
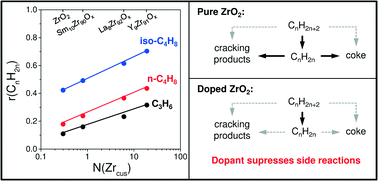
Non-oxidative dehydrogenation (DH) of propane, n-butane, and isobutane was investigated over bare ZrO2 and binary MZrOx (M = Li, Ca, Mg, Y, Sm or La) materials. Selected samples were characterized by XRD, Raman spectroscopy, NH3 temperature-programmed desorption, IR spectroscopy and O2 titration experiments. A correlation between the rate of olefin formation and the concentration of anion vacancies formed upon reductive catalyst treatment was established. Such defects represent the catalytically active sites, which are coordinatively unsaturated Zr4+ (Zrcus) cations. From a structural viewpoint, the active Zrcus sites should have a lower coordination number than the Zrcus sites located on the top of an ideal flat surface, thus highlighting the importance of surface defects in the target reactions. However, the intrinsic activity of such sites and/or their concentration depends on the kind of dopant for ZrO2. The dopant was also shown to inhibit side reactions especially those leading to coke due to reducing the concentration of strong acidic sites related to ZrO2. Similar to an analogue of industrial K-CrOx/Al2O3 catalyst, all binary catalysts exhibited low activity for consecutive reactions of the target olefins; the desired selectivity was higher than 85% at an alkane conversion of about 30%. With respect to the feed alkane, primary selectivity (at a zero degree of alkane conversion) to the target olefin decreases in the order: C3H8, iso-C4H10, and n-C4H10. This was explained by the higher stability of surface intermediates formed from the latter two contributing to the formation of coke and 1,3-butadiene, respectively.


 Please wait while we load your content...
Please wait while we load your content...