Nanobelt α-CuV2O6 with hydrophilic mesoporous poly(ionic liquid): a binary catalyst for synthesis of 2,5-diformylfuran from fructose†
Abstract
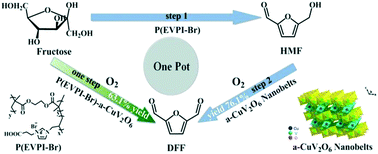
Direct transformation of renewable biomass-derived carbohydrates into value-added chemicals usually involves a tandem reaction, and therefore demands catalysts with different versatile isolated active sites that can independently take action in each step. In this work, the task-specific binary catalyst nanobelt α-CuV2O6 with the mesoporous poly(ionic liquid) (MPIL) derived solid acid P(EVPI-Br) was constructed for the direct synthesis of 2,5-diformylfuran (DFF) from fructose. α-CuV2O6 gave a >99.9% DFF yield in the aerobic oxidation of 5-hydroxymethyl furfural (HMF) under atmospheric O2 (130 °C, 3 h). The binary catalyst α-CuV2O6 with P(EVPI-Br) afforded a 63.1% DFF yield in the one-pot and one-step conversion of fructose into DFF (O2, 135 °C, 3.5 h). Stepwise addition of the two catalyst units further increased the DFF yield to 76.1%. The hydrophilic surface of P(EVPI-Br) enabled the preferential adsorption of fructose on the acid sites whereas α-CuV2O6 showed strong adsorption of HMF but weak affinity to fructose. Such surface wettability-controlled adsorption features inhibited the oxidation of fructose and facilitated the transfer of HMF to the oxidative sites on α-CuV2O6 and its timely conversion into the final product, thanks to the high activity of α-CuV2O6. Moreover, facile catalyst recovery and good reusability were observed in the synthesis of DFF from both HMF and fructose. This work highlights the potential of manipulating the surface wettability of a binary catalyst towards an efficient multifunctional catalysis system for biomass-related tandem reactions.


 Please wait while we load your content...
Please wait while we load your content...