Electrochemically synthesized freestanding 3D nanoporous silver electrode with high electrocatalytic activity†
Abstract
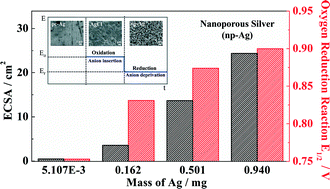
Three-dimensional nanoporous metals of highly porous structure and interconnected ligaments are attractive for electrochemical reactions. Herein, freestanding 3D nanoporous silver (np-Ag) is prepared by a facile electrochemical approach, i.e., first electro-oxidizing silver to silver halides followed by electro-reduction of the silver halides by controlling the potential applied to the electrode. The np-Ag displays 130 and 11.5 times enhancement in catalytic activity for the oxygen reduction reaction and the formaldehyde electro-oxidation reaction, respectively, relative to the flat polycrystalline silver, and even outperforms the commercial nano-Pt catalyst. Detailed experimental and theoretical studies discover that both the facilitated mass transportation in the 3D interconnected porous structures and the favored kinetics contribute to the superior electro-catalytic activity of np-Ag.

 Please wait while we load your content...
Please wait while we load your content...