Local structures and TWC activity of Pd supported on Ni-substituted aluminium oxide borates†
Abstract
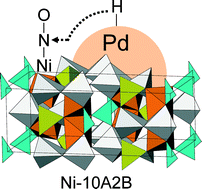
A series of transition metal-substituted aluminium oxide borates, MxAl20−xB4O36 (M–10A2B), were investigated as supports for Pd catalysts. The Ni-substituted compound (Ni–10A2B, x = 1.0) exhibited the highest metal dispersion and catalytic activity under simulated three-way catalysis conditions. X-ray absorption fine structure analysis and density functional theory calculations suggested that Ni2+ occupies an octahedral Al site in the bulk structure. Although Ni2+ ions exposed on the surface of the support are unlikely to contribute to chemisorption under stoichiometric and/or lean conditions, considerable NO adsorption on the Ni sites occurred when the catalyst was partially reduced under rich conditions. Because the as-formed nitrosyl species on Ni–10A2B reacted with hydrogen, which was generated by C3H6–H2O and CO–H2O reactions, and subsequently activated by Pd, the Pd/Ni–10A2B catalyst significantly promoted NO conversion under rich conditions.

 Please wait while we load your content...
Please wait while we load your content...